
These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. The bromide ion, Br–, has a charge of –1. Since CuBr2 is an ionic compound, it forms ions when dissociating in water. Recall that the cathode is where reduction occurs while the anode is where oxidation occurs. We’re being asked to determine the cathode and anode half-reactions for the electrolysis of an aqueous CuBr2 solution. 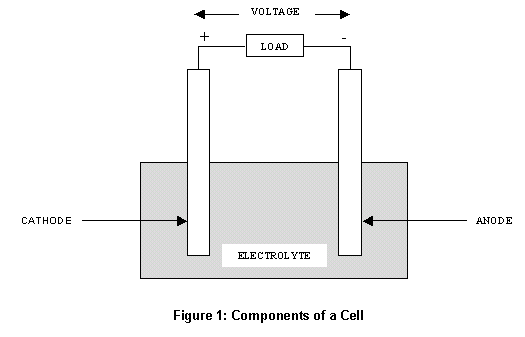
What are the cathode and anode half-reactions for the electrolysis of CuBr2? The metal and hydrogen always forms at the cathode. Oxidation happens at the positive anode because this is where negative ions lose electrons. What type of reaction happens at the anode positive electrode during electrolysis? Br – ions lose electrons at the anode and become Br atoms, which pair up to form Br 2 molecules. During electrolysis: Pb 2+ ions gain electrons at the cathode and become Pb atoms. Molten lead bromide, PbBr 2(l), is an electrolyte. What is produced at anode and cathode during the electrolysis of molten potassium bromide? Thus Ag+(aq) + e- → Ag(s) reaction will be more feasible at cathode. READ ALSO: Can walking a lot hurt your knees? Which of the following equation represent the reaction that takes place at the cathode during the electrolysis of aqueous silver nitrate with carbon electrode? Silver is the cathode (silver ions are reduced). So, in this cell: Zinc is the anode (solid zinc is oxidised). 
How do you write anode and cathode reactions?īy convention in standard cell notation, the anode is written on the left and the cathode is written on the right. Here, the copper ions gain electrons and become solid copper. The half-reaction on the cathode where reduction occurs is Cu2+ (aq) + 2e- = Cu(s). The zinc loses two electrons to form Zn2+. The half-reaction on the anode, where oxidation occurs, is Zn(s) = Zn2+ (aq) + (2e-). What is the half equation for the reaction at the anode? OH – ions are attracted to the anode, lose electrons and form oxygen gas. H+ ions are attracted to the cathode, gain electrons and form hydrogen gas. What are the products formed at anode and cathode? The overall reaction is 2I-(aq) +2H2O(l) –> I2(aq) + H2(g) + 2OH-(aq), the anode reaction is 2I-(aq) –> I2(aq) + 2e- and the cathode reaction is 2H+(aq) + 2e- –> H2(g). READ ALSO: What is the best farm guard dog? What are the anode and cathode reactions for the electrolysis of an aqueous solution of potassium iodide? In pure water at the negatively charged cathode, a reduction reaction takes place, with electrons (e−) from the cathode being given to hydrogen cations to form hydrogen gas. Which reaction takes place at the cathode during the electrolysis of water? They move from anode to the cathode in the external circuit. The electrons are supplied by the species getting oxidized. The reaction at the anode is oxidation and that at the cathode is reduction. 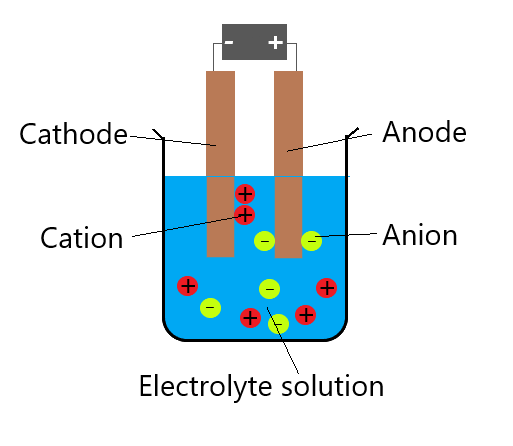
What happens at the anode and cathode in electrolysis?
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
